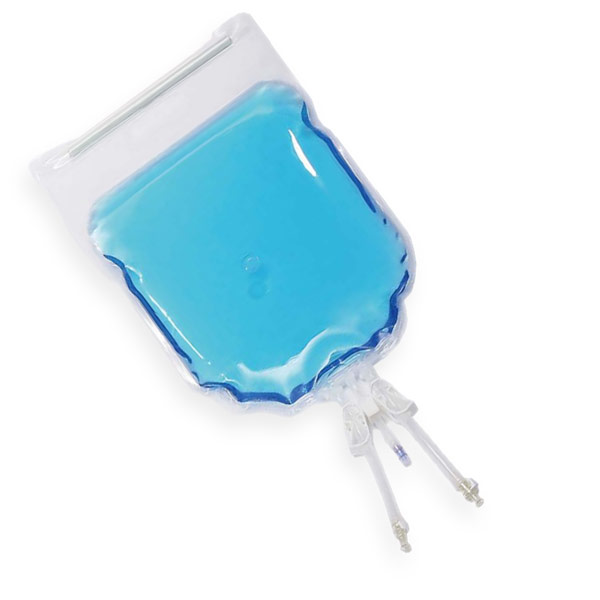
2D Single-use bags
Applicable throughout all process steps in biopharmaceutical production, from 5ml to 50L
Features
- Customizable to various processes
- Low extractables and precipitation
- Gamma-sterilizable
An alternative to bioprocess bags for storage, sampling and transfer and shipping of liquids in all process steps Depending on your production scale, we supply the right size bag for an optimal process.
The 2D Bags from COBETTER are available in the following volumes: 5ml, 50ml, 100ml, 250ml, 500ml, 1L, 2L, 5L, 10L, 20L, 35L, 50L.
If your production requires other volumes or dimensions, ROMYNOX supply a custom made bag based on your specific needs.
Even with small orders, ROMYNOX can assist you. Depending on your needs, we can supply the 2D Bags with different volumes, tubing’s, connectors and assemblies.. The scalability of the 2D Bags allows you to use them throughout the entire biopharmaceutical manufacturing process. Usable from upstream to downstream production (e.g. media and buffer storage, transfer after filtration, storage of intermediates and storage of stock solution).
RENOLIT 9101
Renolit 9101 films are made up of five layers. ULDPE and EVOH prevent the ingress or egress of oxygen, carbon oxide and moisture. LDPE and HDPE guarantee high process reliability. A visual overview of the composition layers is illustrated below.


The 2D single-use bags minimize the risk of cross-contamination and significantly increase safety. Cleaning and sterilization is not required and production efficiency is improved. In addition, it reduces the cost of QC and cleaning verification.
SPECIFICATIONS
- ISO® 9001:2015 quality management system
- ISO class 7 clean zones
- 100% leak test
- ADCF raw materials
- Meet FDA requirements for indirect food additives listed in 21 CFR 177–182
- Meet the requirement of USP <87> In Vitro Biological Reactivity Test
- Meet the criteria of the USP <88> Biological Reactivity Test for Class VI Plastics
- Aqueous extraction contains < 0.25 EU/ml as determined by Limulus Amebocyte Lysate (LAL), USP <85>
- Particles in product eluent meet requirements in USP <788> for large volume parenteral drugs
- Check gamma radiation dose according to ISO® 11137
- Provide a product validation guide and a quality certificate
Read more about Brand: COBETTER

 Single-Use
Single-Use
 Mixers & Pumps
Mixers & Pumps
 Valves
Valves
 Seals
Seals Hoses
Hoses Fittings
Fittings Tube Sealers & Welders
Tube Sealers & Welders
 Transfer Ports
Transfer Ports
 Validation
Validation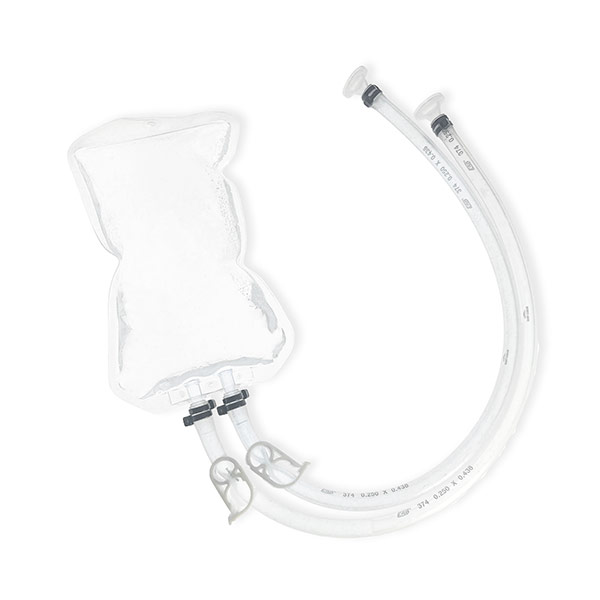




 We utilize cookies to ensure you have the best possible experience while visiting our website.
We utilize cookies to ensure you have the best possible experience while visiting our website.